
Seawater covers approximately 71% of our planet’s surface and plays a crucial role in sustaining life on Earth. One intriguing characteristic that sets it apart from freshwater is its salinity – the presence of dissolved salts in the water. Have you ever wondered why the water in the vast oceans and seas is salty? This question has piqued the curiosity of scientists, explorers, and nature enthusiasts for centuries.
In this article, we will delve into the fascinating reasons behind the salinity of seawater and explore the geological, climatic, and ecological factors that contribute to this unique marine phenomenon. Understanding the origin of the saltiness in seawater will not only enrich our knowledge of the natural world but also shed light on the delicate balance that sustains our marine ecosystems. So, let’s embark on this educational journey to uncover the secrets of why the sea is salty.
Contents
Composition of Seawater.🐟
Seawater is a complex and dynamic mixture of various elements and compounds, making it a unique and essential component of our planet’s ecosystems. Understanding the composition of seawater is fundamental to grasp its role in sustaining marine life, regulating climate, and influencing weather patterns. In this section, we will explore the main components of seawater, delve into its salinity levels and variations across different regions and oceans worldwide, and discuss the presence of other dissolved elements that contribute to its intriguing properties.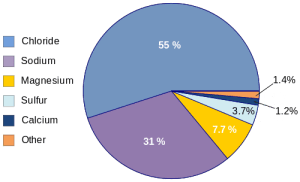
Description of the main components of seawater:
Seawater is primarily composed of two fundamental elements: hydrogen (H) and oxygen (O), forming water molecules (H2O). However, this simple composition accounts for only a small fraction of the overall composition of seawater. The main components are as follows:
- Salts: The most abundant dissolved substances in seawater are salts, predominantly sodium chloride (NaCl). Other salts, such as magnesium, calcium, and potassium, also contribute to the salinity. These salts are derived from various geological processes, such as the weathering of rocks and volcanic activity.
- Gases: Seawater contains significant amounts of dissolved gases, including oxygen and carbon dioxide. These gases play a crucial role in supporting marine life through processes like respiration and photosynthesis.
- Trace elements: Seawater contains trace amounts of numerous elements, including iron, zinc, copper, and manganese. While present in tiny quantities, these elements are vital for the growth and survival of marine organisms.
Salinity percentage and its variation in different regions or oceans:
Salinity refers to the concentration of dissolved salts in seawater, typically expressed as parts per thousand (ppt). The average salinity of seawater is around 35 ppt, meaning that for every 1,000 grams of seawater, approximately 35 grams are dissolved salts.
However, salinity levels can vary significantly depending on various factors, such as evaporation, precipitation, freshwater input from rivers, and ocean currents. Areas with high evaporation rates, such as tropical regions and enclosed seas, tend to have higher salinity levels, while regions near river mouths or experiencing heavy rainfall can have lower salinity levels.
Mention of other dissolved elements in seawater:
In addition to sodium chloride and other salts, seawater contains a rich assortment of other dissolved elements. Some of the notable elements include:
- Carbonates: Seawater contains bicarbonate and carbonate ions, which are essential for the formation of marine shells and coral reefs.
- Nitrogen and Phosphorus: These elements are critical nutrients for marine plant growth and are essential components of the marine food chain.
- Silica: Silica is present in seawater and plays a crucial role in the structure and growth of diatoms, a type of microscopic marine algae
Water Cycle and Salinity.🐟
The water cycle, also known as the hydrological cycle, is a continuous process that circulates water through various stages, including evaporation, condensation, precipitation, and runoff. This natural process plays a significant role in regulating the Earth’s climate, distributing freshwater, and impacting the salinity of the world’s oceans and seas. In this section, we will explore the water cycle’s mechanics and how it influences the salinity of the sea. Additionally, we will describe the key processes of evaporation, precipitation, and runoff in relation to the concentration of salt in the water.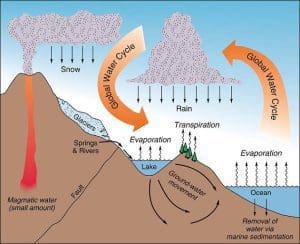
Explanation of the water cycle and its impact on seawater salinity:
The water cycle is a vital process that maintains the Earth’s water balance by continuously cycling water between the atmosphere, land, and oceans. It begins with the process of evaporation, where solar energy heats the surface of oceans, seas, lakes, and rivers, causing water to transform from liquid to water vapor. This water vapor rises into the atmosphere, forming clouds through condensation.
Next, the process of precipitation occurs when the condensed water vapor coalesces and falls back to the Earth’s surface as rain or other forms of precipitation, such as snow or hail. Some of this precipitation directly returns to the oceans and seas, while the rest infiltrates the ground, contributing to groundwater storage or surface water sources like rivers and lakes.
Description of processes in relation to seawater salinity:
As water evaporates from the surface of oceans and seas, the salt and other dissolved substances in seawater are left behind. This process of evaporation concentrates the salt in the remaining seawater, increasing its salinity. Therefore, regions with high rates of evaporation, such as tropical and subtropical areas, tend to have higher salinity levels in their seas.
On the other hand, when precipitation occurs, it brings freshwater to the oceans, diluting the salt concentration. Areas experiencing heavy rainfall or being influenced by freshwater input from rivers typically exhibit lower salinity levels.
Origin of Salinity.🐟
The salinity of seawater is a defining characteristic that distinguishes it from freshwater and plays a crucial role in shaping marine ecosystems. Understanding the origin of this salinity is essential for comprehending the composition of seawater and its significance for life on Earth. In this section, we will explore the general information regarding the sources of salinity in seawater and delve into the accumulation of dissolved salts in the ocean over millions of years.
Main sources of salinity in seawater:
- Rocks: One of the primary sources of salts in seawater is the erosion of rocks on land. As water flows over rocks, it dissolves various minerals, releasing ions such as sodium, calcium, magnesium, and chloride into rivers and ultimately into the ocean. This process is ongoing and contributes significantly to the overall salinity of seawater.
- Soils: The weathering of soils also releases dissolved salts into water bodies. Rainwater leaches minerals from soils, transporting them through rivers to the ocean. The composition of soils in a particular region can influence the type and quantity of salts present in the seawater nearby.
- Volcanic Activity: Underwater volcanic eruptions can release dissolved gases and minerals into the ocean. These minerals, including sulfur and chlorine compounds, contribute to the ocean’s salinity. Additionally, volcanic ash and lava can weather over time, further contributing to the accumulation of salts in seawater.
- River and Oceanic Current Contributions: Rivers carry freshwater from the land to the sea, but they also bring along dissolved salts from their catchment areas. The accumulation of salts in the ocean can be influenced by the balance between the inflow of freshwater from rivers and the outflow through oceanic currents.
Accumulation of dissolved salts in the ocean over millions of years:
The process of accumulating dissolved salts in the ocean has been ongoing for millions of years, ever since the formation of Earth’s oceans. As water from various sources, such as rivers and volcanic activity, carries dissolved minerals and ions, it reaches the oceans. Over time, the concentration of salts in seawater has gradually increased due to the continuous influx and limited removal of these salts from the oceans.
This accumulation is a slow process and occurs over geological timescales. As seawater evaporates, the salts become more concentrated, but they do not evaporate with the water. Therefore, the dissolved salts remain in the ocean, leading to a gradual increase in overall salinity. The balance between processes like evaporation, precipitation, and seawater circulation plays a vital role in determining the final salinity levels in different regions of the world’s oceans.
Effects of Salinity on Marine Ecosystems.🐟
Salinity is a critical factor that profoundly influences marine ecosystems, shaping the distribution and behavior of marine organisms. The varying levels of salt concentration in seawater create diverse habitats with distinct ecological niches. In this section, we will explore the general effects of salinity on marine life and how organisms have adapted to thrive in different saline environments. Additionally, we will highlight some examples of marine species that have evolved unique mechanisms to survive in various salinity levels.
Explanation of how salinity affects marine life and organism adaptation to the saline environment:
Salinity directly impacts marine life by influencing the osmotic balance within organisms. Osmosis is the movement of water across cell membranes, and it is influenced by the concentration of dissolved substances inside and outside of cells. In high-salinity environments, water tends to move out of marine organisms’ cells, causing dehydration and potentially leading to cellular damage. On the other hand, in low-salinity environments, water moves into cells, which can lead to cell bursting.
To cope with these challenges, marine organisms have evolved various adaptations:
- Osmoregulation: Many marine species have sophisticated osmoregulatory mechanisms that help them maintain the proper balance of water and salts inside their cells. For instance, certain fish and invertebrates possess specialized organs, such as salt glands, to excrete excess salts in high-salinity environments.
- Desalination: Some marine plants, such as mangroves, have developed specialized adaptations to desalinate seawater. They can exclude or excrete salt from their tissues, enabling them to survive and grow in brackish or saline conditions.
- Tolerance to Salinity Changes: Some marine organisms, like certain species of jellyfish and algae, have a remarkable ability to tolerate wide variations in salinity. They can thrive in estuaries, where salinity levels fluctuate due to the mixing of freshwater and seawater.
Examples of marine species with salinity adaptation:
- Mangroves (Rhizophora sp.): Mangroves are salt-tolerant trees found in intertidal zones and coastal areas. They have specialized roots and leaf structures that exclude excess salt and allow them to survive in saline conditions.
- Salmon (Oncorhynchus sp.): Salmon are migratory fish that can tolerate varying levels of salinity in their life cycle. They hatch in freshwater rivers, migrate to the ocean as juveniles, and then return to freshwater to reproduce as adults.
- Euryhaline Fish (e.g., Mullet, Flounder): Euryhaline fish are capable of tolerating a wide range of salinity levels. They can be found in both marine and brackish water environments.
- Brine Shrimp (Artemia sp.): Brine shrimp are small crustaceans that inhabit highly saline environments, such as salt flats and salt lakes. They have specialized adaptations to survive in extreme salinity conditions.
Factors Influencing Seawater Salinity.🐟
The salinity of seawater is a dynamic characteristic that can vary across different regions and depths of the ocean. It is influenced by various factors that interact to shape the unique characteristics of marine environments. In this section, we will explore the general factors that influence seawater salinity and delve into the impact of climate and geographic location, as well as the influence of temperature and evaporation on the concentration of salt in the water.
Impact of climate and geographic location on seawater salinity:
- Precipitation and Runoff: Regions experiencing high levels of precipitation and extensive river runoff tend to have lower seawater salinity. Freshwater input dilutes the salt concentration in the ocean. In contrast, arid or desert regions with limited rainfall and minimal river discharge often have higher salinity levels in their surrounding seas.
- Evaporation: Regions with high evaporation rates, such as tropical and subtropical areas, experience increased seawater salinity. As water evaporates from the ocean’s surface, salts and other dissolved substances become more concentrated, leading to higher salinity levels.
- Proximity to River Mouths and Estuaries: The salinity of coastal waters can vary significantly depending on their proximity to river mouths and estuaries. Estuaries, where freshwater from rivers meets seawater, typically have lower salinity levels due to the mixing of waters.
Influence of temperature and evaporation on seawater salinity:
- Temperature: Temperature plays a vital role in the water cycle, which, in turn, affects seawater salinity. Warm temperatures increase evaporation rates, leading to higher salinity in regions with elevated temperatures. Conversely, colder regions may experience lower evaporation rates and, thus, lower salinity.
- Evaporation: Evaporation is a key process that influences seawater salinity. When water evaporates from the ocean’s surface, it leaves the dissolved salts behind, causing the remaining seawater to become more saline.
- Ocean Currents: Ocean currents can transport water with varying salinity levels across different regions of the ocean. Warm, saline currents can increase seawater salinity in some areas, while cold, freshwater currents can lower salinity in others.
Salinity and Climate Change.🐟
Climate change is a global phenomenon that is significantly altering Earth’s climate patterns and affecting various natural processes. One of the critical aspects influenced by climate change is the salinity of seawater. Understanding the potential impacts of climate change on seawater salinity is essential for predicting and mitigating its effects on marine ecosystems. In this section, we will provide general information on how climate change can affect seawater salinity and discuss the potential impacts on marine ecosystems.
Climate change’s potential impact on seawater salinity:
- Changes in Precipitation Patterns: Climate change can lead to alterations in precipitation patterns, including shifts in rainfall frequency and intensity. Changes in precipitation can influence the freshwater input into the oceans, affecting the overall salinity. For example, regions experiencing more intense rainfall may experience a decrease in seawater salinity due to increased freshwater runoff, while arid regions with reduced rainfall may see an increase in salinity.
- Sea Level Rise: Climate change causes the melting of polar ice caps and glaciers, leading to a rise in sea levels. As sea levels rise, coastal areas may experience greater intrusion of seawater into freshwater sources, such as aquifers and river deltas. This seawater intrusion can increase salinity levels in coastal waters and impact nearby ecosystems.
- Ocean Warming and Evaporation: Global warming results in rising ocean temperatures, which can enhance evaporation rates. Increased evaporation can concentrate the salts in seawater, potentially raising its salinity levels in specific regions.
- Melting of Sea Ice: The melting of sea ice in polar regions can influence salinity levels by releasing relatively freshwater into the surrounding seawater. This can create localized areas of lower salinity, impacting marine life that is adapted to specific salt concentrations.
Potential impacts on marine ecosystems:
- Species Distribution and Migration: Changes in seawater salinity can alter the distribution and migration patterns of marine species. Some species may move to areas with more suitable salinity conditions, while others may face challenges in adapting to new salinity levels.
- Impact on Marine Life: Marine organisms, particularly those with specific osmoregulatory requirements, may face physiological stress in response to changing salinity. This stress can affect growth, reproduction, and overall fitness, potentially leading to population declines.
- Ecosystem Dynamics: Altered salinity levels can disrupt the delicate balance within marine ecosystems. Changes in the abundance of certain species can have cascading effects on food chains and ecosystem functioning.
- Loss of Habitats: Rising sea levels and increased salinity in coastal areas can lead to the loss of habitats, such as mangroves and estuaries, which are vital breeding and nursery grounds for many marine species.
Importance of Understanding Seawater Salinity.
Understanding the salinity of seawater is of utmost importance due to its significant impact on marine ecosystems and global climate. Salinity is a fundamental parameter that influences the distribution and adaptation of marine organisms, shapes the characteristics of marine habitats, and plays a crucial role in the Earth’s water cycle. In this article, we have explored various aspects related to seawater salinity, including its composition, origin, and the factors that influence it. Additionally, we discussed the effects of salinity on marine life, adaptation strategies of organisms, and the potential implications of climate change on seawater salinity.
In conclusion, seawater salinity is a complex and dynamic characteristic that significantly influences marine ecosystems and the Earth’s climate system. Understanding its composition, origin, and the various factors that affect it is essential for safeguarding marine life, conserving biodiversity, and addressing the challenges posed by climate change. As stewards of our planet, it is crucial to continue research and collaboration to further our knowledge of seawater salinity and its broader implications. By working together, we can ensure the health and resilience of marine ecosystems for future generations to come.


